Aflatoxin proficiency testing in labs
Timothy Herrman1 and Harinder Makkar2
1 Texas A&M AgriLife Research, College Station TX, USA
2 Food and Agriculture Organization of the United Nations (FAO), Rome, Italy
The use of proficiency testing as a tool to assure the quality of test results is highlighted in the ISO 17025:2005 standard in section 5.9. A proficiency testing programme accompanied by regular use of certified reference material and/or internal quality control material as a secondary reference material represent key components of a laboratory quality system. Proficiency testing programmes enable the evaluation of laboratories for specific tests and may be used by individual labs and their accrediting body to monitor ongoing performance. This aflatoxin proficiency programme provides a useful tool to assist the global feed and food sector to manage aflatoxin risk. Laboratories participate in proficiency testing programmes to identify problems and initiate actions for improvement which may be related to inadequate test or measurement procedures, effectiveness of staff training and supervision, or calibration of equipment. Labs with a Z score over 3 are characterized as not satisfactory and should conduct a corrective action including a root cause analysis to identify the source of variation. Here, we report the approach used and salient findings of the FAO-TAMU Aflatoxin Proficiency Programme, running since last year.
Preparation of proficiency testing samples
Naturally contaminated maize was prepared in 22.68 kg batches from the retained regulatory samples by the Texas Feed and Fertilizer Control Service of the Office of the Texas State Chemist (OTSC). The regulatory samples comprised of 5 kg each, which were sampled using official sampling procedures outline by the United State Department of Agriculture Grain Inspection, Packer and Stockyard Administration (1995) and shipped to the agency’s laboratory in College Station, Texas using a chain-of-custody protocol including double seals and bar codes. Samples were ground with a R.A.S mill from Romer Lab Inc. (Union, MO) and re-ground using a Retsch® Ultra Centrifugal Mill ZM 200 (Haan, Germany) equipped with a 0.75 mm screen. The ground maize was mixed for 1 hour with a commercial Kobalt mixer #0241568 so that it is thoroughly blended. Ground maize was placed into plastic bottles holding approximately 800 g. A reference number created in the laboratory information management system (LIMS) was assigned and labels with bar codes were affixed to each bottle to identify the batch. Bottles were stored in a -20°C freezer and inventory monitored using a log in/log out sheet. The proficiency test item was prepared by placing ground maize into plastic bags until the bag reaches the targeted weight (> 100 g). The plastic bag was then vacuum sealed and labeled for shipment.
The suitability of the reference materials was evaluated using a homogeneity test as follows: for each 22.68 kg batch, 12 bottles of the prepared maize were randomly selected and 50 ± 0.25 g samples were weighed in duplicate from each of the 12 bottles. Homogeneity evaluation was performed using ISO/IEC 13528:2015 statistical methods for use in proficiency testing by inter-laboratory comparisons found in Annex B, B.3 using the formulae for homogeneity check. The stability test compared 5 samples (vacuum sealed plastic bags) retained in the proficiency test item production and the 5 samples that are retrieved from the stability challenge. A t-test for significant difference at the 95% level of confidence was used to evaluate the stability test results.
The aflatoxin testing procedures conformed to the AOAC methodology for high performance liquid chromatography (HPLC), UV and florescence detection (AOAC 2005). Certified AFB1, AFB2, AFG1, and AFG2 were purchased from Romer Labs-Biopure (Tulln, Austria) and used as standards. All solutions are made with HPLC-grade solvents and reagent grade materials. The concentration of AFB1 and AFG1 standard is 2 µg/mL in 5 mL acetonitrile. The concentration of AFB2 and AFG2 standard is 0.5 µg/mL in 5 mL acetonitrile.
Proficiency testing programme registration, sample delivery, and reporting
Participants registered for the aflatoxin proficiency testing programme in response to direct email contact from the Food and Agriculture Organization (FAO) of the United Nations, through open solicitation in trade magazines, and through a several laboratory networks established by Texas A&M AgriLife Research in collaboration with the Kenya milling industry and the Common Market for Eastern and Southern Africa (COMESA). Registration included identification of the contact person, laboratory name, mailing address, phone and fax number and contact email address. The registrants also designated if they were a grain miller, grain handler, feed company, government or research entity as well as the testing platform including manufacturer name and model if high performance liquid chromatography or the name of the test kit and reader if the later was used. Samples were shipped via express courier and all packages were tracked by the shipper. Accompanying the samples were instructions to the laboratory involving sample analysis and data entry procedure. Laboratories were instructed to analyze samples twice and enter the data into the populated website with their laboratory information and respective laboratory code. Participant identification was protected by a lab code, user name, and password. A final data entry date was assigned and submissions were not accepted afterwards. The reports for round one and two were automatically generated online within 2 days of the final submission date and were accessible to all participants through the electronic porthole accessible using the same login information. Reports contained elements required in the ISO 17043 standard including the participant’s results, statistical data and summaries including assigned values and range of acceptable results and graphical displays (Figure 1).
Proficiency test result analysis and interpretation
The assigned value for the measurand (aflatoxin) was based on aflatoxin analysis results following the procedures outlined above. The evaluation of the participants using the Z score calculation was calculated based on the following equation:
z = (x – μ) / σ
where z is the Z score to evaluate the individual laboratory performance, x is the mean value of the two individual laboratory reported numbers, μ is the assigned value for the corresponding PT item, and σ is calculated based on the following equation:
σ = {Cx2^[1 - log(C)/2]}/100
where C is the concentration of aflatoxin.
Z score evaluation was as follows:
│z│ ≤ 2.0 satisfactory
2.0 < │z│ < 3.0 questionable
│z│ ≥ 3.0 not satisfactory
The Cochran test eliminated outliers with high intra-lab variability and Grubbs test eliminated outliers based on between laboratory variability from the composite statistical analysis.
Global aflatoxin testing performance
Round One: Ninety laboratories received aflatoxin proficiency samples and a total of 84 laboratories submitted data through the programme’s electronic porthole. Results showed that data from 96% of the laboratories were acceptable, one result was eliminated using the Cochran test and two lab results were eliminated using the Grubbs test as outliers. The participant mean results were slightly higher (4 parts per billion) than the assigned value and the average variability of the test results were double that of the proficiency provider’s homogeneity test results. These are both indicators that the proficiency programme successful met criteria for a proficiency testing programme
Round Two: A total of 114 laboratories participated in the second round of 2016 aflatoxin proficiency test, of which 61 laboratories analyzed for both B1 toxin and total. The remaining 53 laboratories tested for either B1 or total aflatoxin. Laboratories participated from 5 continents with 55 from Africa, 21 Asia, 22 Europe, 9 North America and 5 South America. A total of 175 results were entered of which 48 results were from test kits, 91 from liquid chromatograph, 24 used liquid chromatography with mass spectrometry, and 12 used other testing platforms such as thin-layer chromatography.
Key performance statistics include the mean, range, bias, and Z score. The assigned mean for total aflatoxin was 34.5 µg/kg while the participants’ average was 32.8 µg/kg. The assigned standard deviation for total aflatoxin using the Horwitz equations was 9.2 µg/kg and the participants’ standard deviation was 12.2 µg/kg aflatoxin. Samples were measured in duplicate and the Cochran test of the variance of results from each lab removed one outlier. The Grubbs test of the laboratory means showed no outliers. For B1 aflatoxin, the assigned mean was 31.8 µg/kg while the reported mean was 27.6 µg/kg and the assigned standard deviation of 8.5 µg/kg and the reported standard deviation was 12.3 µg/kg. For B1, there were no outliers.
Five of the Z scores for total aflatoxin were greater than 3. For B1, 7 of the absolute Z scores were great than 3. A score of zero implies a perfect result, approximately 95% of z-scores fall between -2 and +2, and a score outside the range from -3 to 3 should be investigated and accompanied by a corrective action. While only three results from the first round had Z scores greater than 3, the number of laboratory results from the first round were approximately half compared to the second round. The composite relative standard deviation was 37% in the second round, slightly higher than the 35% reported than the first round.
A comparison between continents and testing platforms for total aflatoxin and B1 aflatoxin was performed. African laboratories reported total aflatoxin using test kit platforms with an average relative standard deviation of 33.6%, a composite mean 33.9 µg/kg, and a composite bias of -0.08, the lowest among the continent groupings. Seventeen of 20 laboratories had Z scores less than 2 and none of the Z scores were greater than 3. The Europe laboratories performed equally well for aflatoxin B1 using liquid chromatography with an average relative standard deviation of 34.8%, a mean of 31.3 µg/kg and a bias of 0.23 µg/kg.
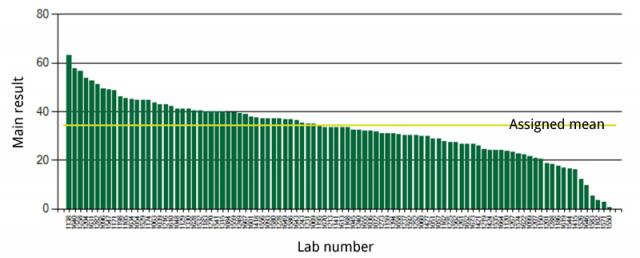
Figure 1. Total aflatoxin results among labs participating in the second round.
Summary
This proficiency programme highlights the global aflatoxin testing capability. It also reflects the priority in testing and managing aflatoxin risk in different regions of the world. For example, several activities including the Aflatoxin Proficiency Testing and Control in Africa (APTECA) programme by Texas A&M AgriLife Research, the Laboratory Quality Systems online course offered by Texas A&M in collaboration with FAO, and several other FAO steps taken to assist adoption of good laboratory practices and laboratory quality control systems (FAO, 2016) contributed to the quality test results by participants from previously established laboratory networks.
Literature cited
AOAC (Association of Official Analytical Chemists. (2005). Aflatoxins in corn, raw peanuts and peanut butter, liquid chromatography with post-column photochemical derivatization. Official method 2005.08, 49.2.18A
FAO (2016). FAO assistance towards feed analysis: increasing income, improving food safety and safeguarding the environment, FAO, Rome, Italy. Available at: http://www.fao.org/publications/card/en/c/c4d482cb-14b6-4d46-879e-4dd580920077/
ISO/IEC 17025, General requirements for the competence of testing and calibration laboratories
ISO 13528:2005, Statistical methods for use in proficiency testing by interlaboratory comparisons
ISO 17034:2010. Conformity assessment – General requirements for proficiency testing
USDA-GIPSA (U.S. Department of Agriculture Grain Inspection, Packers, and Stockyards Administration). (1995). Probe sampling. In Grain inspection handbook, Chapter 2. (pp. 1-20). Federal Grain Inspection Service. Washington, DC. Available at https://www.gipsa.usda.gov/fgis/handbook/BK1/BookI_2015-09-18.pdf

