NIR-based detection of contaminants in food and feed
V. Baeten, P. Dardenne
Walloon Agricultural Research Centre, Gembloux, Belgium
Contact: v.baeten@cra.wallonie.be, FoodFeedQuality@cra.wallonie.be
The use of near-infrared (NIR) technologies for the detection of contaminants and undesirable substances in food and feed products is not widely practised. There have been many papers, however, on extensive studies on this topic (see Baeten et al., 2015). They have demonstrated some unique advantages of using this fingerprinting technique in the continuing effort to give the stakeholders the means to detect contaminants at all stages of the food and feed chains. This is crucial for the common objective of these chains, which is to provide products that do not adversely affect animal and human health. For more than 15 years, the Walloon Agricultural Research Centre (CRA-W) has helped to demonstrate the potential of NIR spectroscopy in the detection, identification and quantification of animal, plant and chemical contaminants and undesirable substances.
Near Infrared spectroscopy and optical microscopy
This work has been based on several NIR technologies (i.e., classical NIR, NIR microscopy, NIR hyperspectral imaging, on-line NIR and handheld NIR instruments) and is a synthesis of more than 40 scientific papers and book chapters.

Photo: Fresh forage analysis using a portable micro-NIR instrument
The achievements so far show that NIR, combined with adequate sample presentation and appropriate chemometric tools, has a specific place in strategies to ensure the analytical control of contaminants at the farmer, industry and laboratory levels. This needs to be done through the implementation of appropriate solutions in terms of limit of detection (LOD), robustness and sampling issues.
The first study on contaminant detection using NIRM (Near InfraRed Microscopy) was conducted at CRA-W in 1998. It focused on the application of the NIRM technique to the detection of forbidden ingredients in animal feed. Within the framework of the mad cow disease crisis, our objective was to develop a method that could detect animal ingredients in compound feed. Processed animal protein (PAP) had been forbidden in an effort to stop the spread of bovine encephalopathy in the European herd. In order to reach the required LOD (0.1%), it was decided to use an NIR spectrometer coupled with a microscope (NIRM instrument) (Baeten and Dardenne, 2001). This instrument enabled us to collect NIR spectra of individual particles in order to determine if they were from animal, plant or mineral origin. Figure 1 presents a plant spectrum and an animal spectrum extracted from a feed sample.

Figure 1: NIR spectra of vegetal and animal feed particles in the 1100-2500 nm range
The studies demonstrated the potential of NIRM by developing a methodology for detecting animal ingredient particles in compound feed. In order to improve the sensitivity and specificity of the NIRM method, a hierarchical combination of partial least squares discriminant analysis (PLS-DA) models were proposed, which enabled to achieve a correct classification rate of 99.9% (Baeten et al., 2004).
Near InfraRed HyperSpectral Imaging (NIR-HSI)
The time required (2-3 days), however, for collecting the NIR spectra of the 3,000 particles, which had to be analysed to reach the required LOD in compound feed, was far too long (Abbas et al., 2010; Yang et al., 2011). In order to tackle this problem, two options were investigated: introduction of a step to concentrate targeted particles; and use of an NIR hyperspectral instrument. The first option, implemented by introducing a sedimentation step into the NIRM analytical protocol, in order to concentrate the bone fragments in the heavy fraction, enabled to reach the required LOD and to reduce the analytical time by a factor about 5 (Baeten et al., 2005). This protocol was compared with the official method (classical microscopy) on a batch of blind spiked samples. A correlation coefficient of 0.93 between both methods showed that the sedimentation step was efficient. The second option was implemented by using an NIR hyperspectral imaging system (NIR-HSI) and discrimination models based on support vector machine (SVM) (Fernández Pierna et al., 2004). This option enabled to reduce the analytical time by a factor about 20, achieving the required LOD and without needing a sedimentation step. In addition, the studies conducted using NIRM and NIR-HSI instruments demonstrated a potential to discriminate, to some extent, the species group origin of the animal particles (e.g., terrestrial origin vs fish origin) (Baeten et al., 2004; de la Haba et al., 2007). The compounds and the related NIR bands were identified in several studies (Tena et al., 2014). The discrimination power of the method, however, had limitations and it could not be seen as a species identification method. An interesting approach to overcome this limitation was to combine the NIRM protocol with a polymerase chain reaction (PCR) protocol that would detect the animal origin of the particle and then identify its species origin by DNA analysis (Fumière et al., 2010). The good performance, repeatability and speed of the NIR based methodologies for detecting animal particles led to the use of these methods in a homogeneity study of the materials used in European inter-laboratory studies aimed at testing the ability of the European Member States to implement the official method (classical microscopy) for detecting PAP in feed. The methodologies are also being used in the ISO17025 accreditation scheme (Fernández Pierna et al., 2010). More recently, a protocol based on using a standardisation cell specifically designed to standardise NIR microscopes has been successfully developed (Fernández Pierna et al., 2013). Within the framework of the Safeed-PAP project, the NIRM protocol has been tested by eight laboratories and has been validated according to international standards (Boix et al., 2012). The potential of NIRM and NIR-HSI methods, however, is limited in terms of quantifying animal protein in compound feed; they can be considered only as semi-quantitative methods. This is probably due mainly to the sedimentation step which does not take into account the difference in size and density of different types of particles.
NIRM and NIR-HSI techniques have been used not only for detecting animal particles in compound feed, but also for developing methods for the full screening of particles in ground compound feed. They have been also proposed for detecting other types of plant and mineral contaminants (Baeten et al., 2010; Fernández Pierna et al., 2012). The NIR imaging system has been used successfully to detect, at the ppm level, the presence of ergot bodies and other plant contaminants and the botanical impurities in cereals. One study has shown that NIR-HSI technology can be used to detect and quantify ergot contamination in cereals destined for food or feed, in both the laboratory and the production chain, using online prediction (Vermeulen et al., 2012). The NIR-HSI method enables a 250 g sample to be analysed in 1 minute using the push-broom (moving belt) HSI system. It was assessed that using a set-up based on the full conveyor belt width (30 cm) and a belt speed of 100 mm/s, up to about 100 kg grain/hour could be analysed, whereas the current official microscopy method requires 15-60 min to analyse 250 g. The protocol was successfully transferred from a pilot system used at laboratory level to a commercial system tested at industrial level and showed that the NIR-HSI could be a powerful tool within the framework of official controls (Vermeulen et al., 2015). Such a method could also be useful as an analytical screening tool in an automatic cereal control scheme, where the positive samples could be analysed using official methods.
Melamine crisis
The melamine crises (2007, 2008, 2009) also led to proposals to use NIR techniques (NIRS, NIRM and NIR-HSI) to detect melamine, first in feed and later in food (i.e., milk and milk powder).
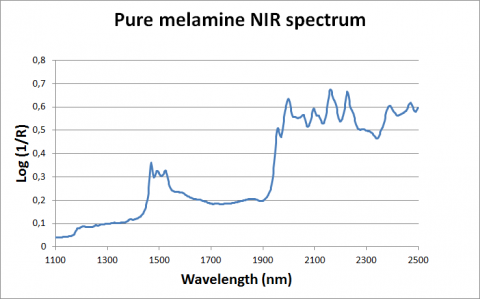
Figure 2: NIR spectrum of pure melamine in the 1100-2500 nm range
These crises, linked to animal deaths and human health, demonstrated the need for a sensitive, reliable and rapid procedure for determining the presence of melamine and other contaminants in agro-food products (Abbas et al., 2013; Fernández Pierna et al., 2014). The challenge, however, is to be able to detect the next crisis (Baeten et al., 2014). In order to achieve this, CRA-W has proposed, within the framework of the EU’s QSAFFE project (http://www.qsaffe.eu/), a procedure based on NIR spectroscopy and a combination of chemometric steps in order to characterize soybean meal and to detect the presence of any known or unknown contaminants before they reach the feed chain (Fernández Pierna et al., 2015). The detection can be based using 3 tools: 1) the prediction of classical constituents or contaminants using quantification models, 2) the use of generalized distances between an unknown and a clean data set, and 3) the spectral residuals between the checked sample spectrum and a selected subset of clean sample spectra. This procedure allows compliance to be checked against specifications in order to decide whether to reject or accept a product (Fernandez Pierna et al., 2015).
Very low level analytes
There is anyway a big obstacle to detect contaminant by NIR using a global sample presentation (one averaged spectrum/sample): NIR, in most of the feed and food matrices, is unable to detect very low concentrations such as those of antibiotics or pesticide residues present in feeds. The limits of detection, depending on the molecular absorptivities of the searched molecules, will be between 200 and 500 ppm (0.02 - 0.05%). Above these limits of concentration, the potential of NIR technologies in the detection of contaminants is obvious.
The future
With instrument miniaturization and the reduction of the instrument costs, it is clear that they will play an increasingly important role in ensuring safety in the feed and food chains. The drawback of “global” NIR (one spectrum/sample) can be overcome with NIR microscopy and NIR-HIS. These techniques are capable of acquiring thousands of spectra for each sample. Depending on the particle size and distribution, spectra can be produced by “pure” products inside the matrix allowing lower limits of detection. NIR and NIR-HIS can be easily implemented, and as the cost of the sensors is going down, they will appear more and more not only in the industrial and agricultural worlds but also in our consumer life. Connected through a smart phone to specialized servers, the instrument will give instantly several quality parameters on any scanned matter.
Acknowledgement
The authors thank the staff of the Food and Feed Quality Unit of CRA-W for their expertise demonstrated in the framework of the different studies mentioned in this paper.
References
Abbas, O. ; Fernández Pierna, J. A. ; Boix, A.; von Holst, C.; Dardenne, P.; Baeten, V. (2010). Key parameters for the development of a NIR microscopic method for the quantification of by-products of animal origin in compound feedingstuffs. Analytical; Bioanalytical Chemistry, 397, 1965-1973.
Abbas O.; Lecler B.; Dardenne, P.; Baeten, V. (2013) Detection of melamine in feed ingredients by near infrared spectroscopy; chemometrics. Journal of Near Infrared Spectroscopy, 21(3), 183-194.
Baeten, V.; Dardenne, P. (2001). The contribution of near infrared spectroscopy to the fight against the mad cow epidemic. NIRS news, 12(6), 12-13.
Baeten, V.; Fernández Pierna, J. A.; Lecler B.; Abbas O.; Vincke D.; Minet, O.; Vermeulen, Ph.; Dardenne, P. (2016) Near infrared spectroscopy for food; feed: a mature Technique.NIRS news, 27 (1), 4-6.
Baeten, V.; Vermeulen, Ph.; Fernández Pierna, J. A.; Dardenne, P. (2014). From targeted to untargeted detection of contaminants; foreign bodies in food; feed using NIR spectroscopy. New Food, 17(3), 16-23.
Baeten, V.; von Holst C.; Garrido A.; VanCutsem, J.; Michotte Renier, A.; Dardenne, P. (2005). Detection of banned Meat; Bone Meal in Feedingstuffs by Near-infrared Microscopy Analysis of the Sediment Fraction. Anal. Bioanal. Chem., 382, 149-157.
Baeten, V. ; von Holst, C. ; Fissiaux, I.; Michotte Renier, A.; Murray, I.; Dardenne, P. (2004). The near infrared microscopic (NIRM) method: combination of the advantages of optical microscopy; near-infrared spectroscopy (WP5-NIRM). In: Strategies; methods to detect; quantify mammalian tissues in feedingstuffs, Bruxelles - Belgium, European Commission.
Baeten, V.; Rogez, H.; Fernández Pierna, J. A.; Vermeulen, Ph.; Dardenne, P. (2015). Vibrational Spectroscopy Methods for the Rapid Control of Agro- Food Products. In Handbook of Food Analysis (3rd Edition). (Eds. Toldra; Nollet). Volume II, Chapter 32, pp. 591-614.
Boix, A. ; Fernández Pierna, J. A. ; Von Holst, C.; Baeten, V. (2012). Validation of a near infrared microscopy method for the detection of animal products in feedingstuffs: Results of a collaborative study. Food Additives; Contaminants Part A.; 29 (12), 1872-1880.
de la Haba, M. J.; Fernández Pierna, J. A.; Fumière, O.; Garrido-Varo, A.; Guerrero J.E.; Pérez-Marín D.C.; Dardenne, P.; Baeten, V.; Discrimination of fish bones from other animal bones in the sedimented fraction of compound feeds ny near infrared microscopy (NIRM). (2007). J. of NIRS, 15, 81-88.
Fernández Pierna, J. A.; Abbas, O.; Lecler, B.; Hogrel, P.; Dardenne, P.; Baeten, V. (2015). NIR fingerprint screening for early control of non-conformity at feed mills. Food Chemistry, 189, art. no. 16461, pp. 2-12.
Fernández Pierna, J. A.; Baeten, V.; Michotte Renier, A.; Cogdill, R.P.; Dardenne, P. (2005). Combination of SVM; NIR imaging spectroscopy for the detection of MBM in compound feeds. Journal of Chemometrics, 18(7-8), 341-349.
Fernández Pierna, J. A.; Boix, A.; Slowikowski, B.; von Holst, C.; Maute, O.; Han, L.; Amato, G.; de la Roza Delgado, B.; Marín, D.; Lilley, G.; Dardenne, P.; Baeten, V. (2013). Standardization of NIR microscopy spectra obtained from inter-laboratory studies by using a standardization cell. Revue de Biotechnologie, Agronomie, Société et Environnement,17(4), 547-555.
Fernández Pierna, J. A.; Dardenne, P.; Baeten, V. (2010). In-house validation of a near infrared hyperspectral imaging method for detecting processed animal proteins (PAP) in compound feed. Journal of Near Infrared Spectroscopy, 18, 121-133.
Fernández Pierna, J. A.; Vermeulen, P.; Amand, O.; Tossens, A.; Dardenne, P.; Baeten, V. (2012) NIR hyperspectral imaging spectroscopy; chemometrics for the detection of undesirable substances in food; feed. Chemometrics; Intelligent Laboratory Systems, 117, 233-239.
Fernández Pierna, J. A.; Vincke, D.; Dardenne, P.; Yang, Z.; Han, L.; Baeten, V. (2014). Line scan hyperspectral imaging spectroscopyfor the early detection of melamineand cyanuric acid in feed. Journal of Near Infrared Spectroscopy, 22 (2), pp. 103-112.
Fernandez Pierna, J. A. ; Vincke, D. ; Baeten, V. ; Grelet, C. ; Dehareng, F. ; Dardenne, P. (2016). Use of a multivariate moving window PCA for the untargeted detection of contaminants in agro-food products, as exemplified by the detection of melamine levels in milk using vibrational spectroscopy. Chemometrics; intelligent laboratory systems, 152, 157-162.
Fumière, O.; Marien, A.; Fernández Pierna, J. A.; Baeten, V.; Berben G. (2010). Development of a real-time PCR protocol for the species origin confirmation of isolated animal particles detected by NIRM. Food Additives; Contaminants, 27(8), 1118-1127.
Tena, N.; Fernández Pierna, J. A.; Boix, A.; Baeten, V.; von Holst, C. (2014). Differentiation of meat; bone meal from fishmeal by near-infrared microscopy: Extension of scope to defatted samples. Food Control, 43, 155-162.
Vermeulen, P.; Fernandez Pierna, J. A.; van Egmond, H. P.; Dardenne, P.; Baeten, V. (2012). Online detection; quantification of ergot bodies in cereals using near infrared hyperspectral imaging. Food additives; contaminants, 29(2), 232-40.
Vermeulen, Ph.; Fernández Pierna, J. A.; van Egmond, H.P.; Zegers, J.; Dardenne, P.; Baeten, V. (2013). Validation; transferability study of a method based on near-infrared hyperspectral imaging for the detection; quantification of ergot bodies in cereals. Analytical; Bioanalytical Chemistry, 405 (24), 7765-7772.
Yang, Z.; Han, L.; Fernández Pierna, J. A.; Dardenne, P.; Baeten, V. (2011). Review of the potential of near infrared microscopy to detect, identify; quantify processed animal by-products. Journal of NIRS, 19(4), 211-231.